Research
The main topic of the research group is the application of high pressure NMR spectroscopy to bio(macro)molecules and their use for drug design. High pressure NMR spectroscopy is a novel method to study rare (“excited”) states of macromolecules with a sensitivity down to the ppm range. Rare excited states have a higher free Gibbs energy DG than the ground state and thus a lower relative occupancy at ambient pressure. At high pressure the occupancies of the states can be inversed because of differences in the partial molar volumes DV of the states and thus become directly detectable by NMR spectroscopy. We have built different high pressure systems that allow application of pressures up to 300 MPa and pressure jumps as fast as 100 us/MPa in the NMR spectrometer (300 MPa would correspond to the pressure 30 000 m below sea level).
Using this method we could define three conformational states of the peptide Ab that are involved in the development of Alzheimer disease. It is assumed that only one of the conformational states id competent for fibril formation (Fig. 1).
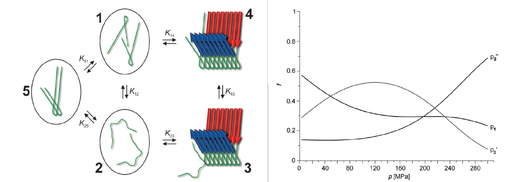
Fig. 1. Different conformational states in Alzheimer Amyloid A detected by high pressure NMR spectroscopy (from Munte et al. (2013) Angew. Chem. Int. Ed.). (left) model of fibril formation (right) Dependence of population f of different states from pressure p.
Mutations in the small G-protein Ras are found in about 30 % of all human tumors. It occurs in its inactive state when the nucleotide GDP is bound to its active center and transmits a proliferation signal when GDP is exchange to GTP by a guanine exchange factor GEF. It is switched off by a GTPase activating protein GAP (Fig. 2). High pressure NMR spectroscopy allowed to detect 4 conformational states of activated Ras protein in the cycle. Stabilization of one of the states (state 1(T)) by Zn-cyclen or Zn-BPA leads to an allosteric inhibition of Ras.
Other applications performed in the group concern NMR based metabolomics in body fluids and living cells as well as software development for NMR spectroscopy.

Fig. 2 Ras cycle and conformational states allowing the development of allosteric inhibitors. (from Kalbitzer et al (2014) and Rosnizeck et al- 2013) Angew. Chem. Int. Ed.
Publications
See pubmed
CV
Education:
1967-1971 Study of Medicine and Physics, University of Göttingen
1971-1976 Study of Medicine and/or Physics, University of Heidelberg, “Staatsexamen“ in Medicine 1973, Diploma in Physics 1976
1976 Dr. med, (M.D.) University of Heidelberg
1978 “Approbation als Arzt” (Licence to practice)
1981 Dr. rer. nat. (Ph.D.), University of Heidelberg (Physics)
1984 Habilitation and Venia Legendi in Biochemistry, University of Regensburg, Germany
1991 Apl. Prof. of Biochemistry
1995 Venia legendi in Biophysics
1996 "Facharzt" for Biochemistry
Positions:
1977-1986 Research Associate, Max-Planck-Institute for Medical Research, Dept.
Molecular Physics, Heidelberg
1983 Visiting Scientist, Stanford University, Palo Alto
1986-1997 Senior Scientist, Max-Planck-Institute for Medical Research, Dept.
Biophysics, Heidelberg
1997 Visiting Scientist, Kobe University, Kobe
1997-2014 Full professor (C4), Institute of Biophysics and Physical Biochemistry, Chair for Biophysics, University of Regensburg
2000/2001 Visiting Scientist, CNB-CSIC, Protein Design Group, Cantoblanco,
Spain
2008/2009 Visiting scientist, UC Berkeley, Berkeley
Since 2014 Senior professor at the University of Regensburg. High Pressure NMR Spectroscopy of Biomolecules


