
Research
Our group uses and develops dyes and luminescent probes for the development of bioanalytical assays and sensors. Those dyes may indicate the presence of an analyte by appearance of color (i.e. chromogenic dyes) or by a color change. Both types of dyes may be used in photometric assays and for the reflectometric readout of test stripes. We further create probes that respond to the presence of an analyte by changes of the various parameters of luminescence. All those probes serve to render biomolecules (e.g. hydrogen peroxide, amines, DNA, ions, enzymes, enzyme substrates/products, pH, gases and tumor metabolites) amenable for quantitation with optical readout. We are further involved in food analysis i.e. in the development of new methods for optical quantitation of biogenic amines.
Prominent Topics
Test Stripes
Test stripes should deliver a rapid yes/no-answer or a semiquantitative result to the layman. We create such stripes by embedding suitable probes into polymer matrices that are coated onto a solid support. The picture below shows the response of test stripes to biogenic amines (BAs). Those are indicators of the freshness of protein-rich food because they are formed by enzymatic degradation upon ageing of food like fish, meat and cheese. Increasing concentrations of BAs are displayed by a red color. The stripes were designed to respond to BA-levels that are already toxic (and hence can induce food poisoning) but are below the odor threshold of the human nose.

Further analytes relevant for new test stripes are alcohols, aldehydes, ketones, pH and heavy metal ions. We employ microplate readers, spectrometers, digital cameras, mobile phones and digital pocket microscopes for the evaluation of stripes.
Selected publications:
- Yurova NS, Danchuk A, Mobarez SN, Wongkaew N, Rusanova TY, Baeumner AJ, Duerkop A, 2018, Functional Electrospun Nanofibers for Multimodal Sensitive Detection of Biogenic Amines in Food via a Simple Dipstick Assay, Anal Bioanal Chem, 410:1111-1121, DOI: 10.1007/s00216-017-0696-9.
- Bidmanova S, Steiner M-S, Stepan M, Vymazalova K, Gruber MA, Duerkop A, Damborsky J, Prokop Z, Wolfbeis OS, 2016, Enzyme based test stripes for the visual or reflectometric detection and quantitation of gaseous sulfur mustard, Anal Chem 88:6044-6049, DOI: 10.1021/acs.analchem.6b01272
- Steiner M-S, Meier, RJ, Duerkop A, Wolfbeis OS, 2010, Chromogenic Sensing of Biogenic Amines Using a Chameleon Probe and the Red Green Blue Readout of Digital Camera Images, Anal Chem, 82:8402-8405, doi: 10.1021/ac102029j
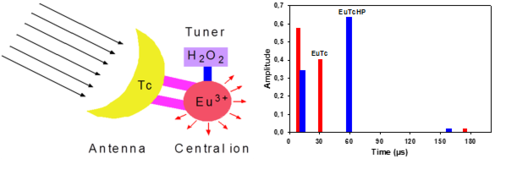
Probes for Hydrogen Peroxide
Hydrogen peroxide (HP) has a central role as second messenger and reaction product of enzymatic and metabolic activity. On the other hand, HP is a reactive oxygen species and cell toxic. Therefore, the oxidative stress of cells may be monitored by probes for HP via the activity of oxidases. Hence, we develop new luminescent organic dyes and lanthanide complexes for probing HP via intensity and luminescence lifetime. Those are used either in microplate assays (for high sample throughput) but as well in sensors for the continuous measurement of HP. Green chemistry is addressed by creating sensor microplates for repeated use to reduce the waste of bioanalytical labs.
Selected publications:
- Burmistrova NA, Kolontaeva OA, Duerkop A, 2015, New Nanomaterials and Luminescent Optical Sensors for Detection of Hydrogen Peroxide, Chemosensors 3:253-273, doi:10.3390/chemosensors3040253
- Burmistrova NA, Meier RJ, Schreml S, Duerkop A, 2014, Reusable Optical Sensing Microplate for Hydrogen Peroxide Using a Fluorescent Photoinduced Electron Transfer Probe (HP Green), Sens Actuat B 193:799-805, doi: 10.1016/j.snb.2013.12.025.
- Wolfbeis OS, Dürkop A, Wu M, Lin Z, 2002, Europium Ion-Based Luminescent Sensing Probe for Hydrogen Peroxide, Angew Chem Int Ed, 41:4495-4498
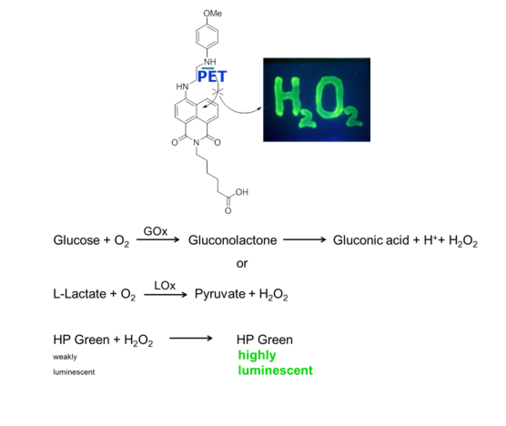
Enzymatic Luminescence Assays
Oxidases convert their substrates under the release of HP. The HP level is directly related to the amount of substrate that is present in the sample or the amount of product that has been formed. Hence, concentrations of substrates or products of oxidases can be quantified by probes for HP. This opens access to a variety of bioanalytes like glucose, lactate, uric acid, sarcosine, superoxide radicals, xanthine, alcohols and amines.
Selected publications:
- Groegel DBM, Link M, Duerkop A*, Wolfbeis OS, 2011, A New Fluorescent PET Probe for Hydrogen Peroxide and its Use in Enzymatic Assays for L-Lactate and D-Glucose, ChemBioChem, 12:2779-2785, doi: 10.1002/cbic.201100561
- Wu M, Lin Z, Schäferling M, Dürkop A, Wolfbeis OS, 2005, Fluorescence Imaging of the Activity of Glucose Oxidase Using a Hydrogen Peroxide Sensitive Europium Probe, Anal Biochem, 340:66-73.
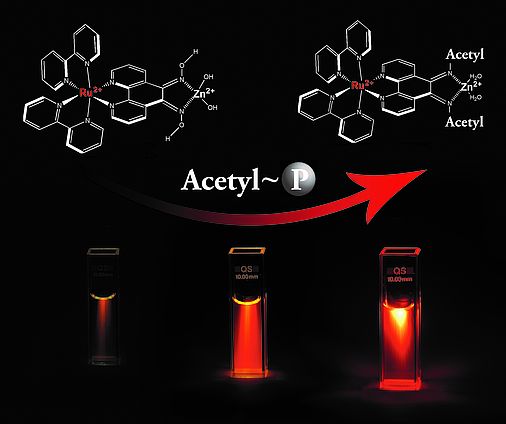
Luminescent Complexes for Bioanalysis
Transition metal-ligand complexes und lanthanide complexes are promising luminescent molecules for bioanalysis. We develop ruthenium complexes for bioconjugation to amines or thiol groups in biomolecules, as DNA-intercalators and as probes for polarization immunoassays. These complexes may also be used for quantitation of saccharides or tumor markers or pH. Lanthanide complexes are well soluble in biological fluids and show narrow emission bands and long luminescence decay times. We therefore use them for ion analysis (e.g. Cu(II), phosphate, citrate), DNA, pesticides and for the screening for antibiotics.
Selected publications:
- Waleed Al-Qaisy W, Duerkop A, 2018, Luminescent europium complex for wide-range pH sensor and sensor microtiterplate, Analyst, 143, 3176 – 3183, DOI: 10.1039/C8AN00775F
- Kele P, Li X, Duerkop A, 2013, Luminescent Ruthenium Complexes for Diacetyl Detection, Microchem J 108:156–160, doi:10.1016/j.microc.2012.10.012.
- Steiner M-S, Duerkop A*, 2011, Luminescent Ruthenium Probe for the Determination of Acetyl Phosphate in Complex Biological Matrices, Analyst, 136:148-154, doi: 10.1039/C0AN00439A.
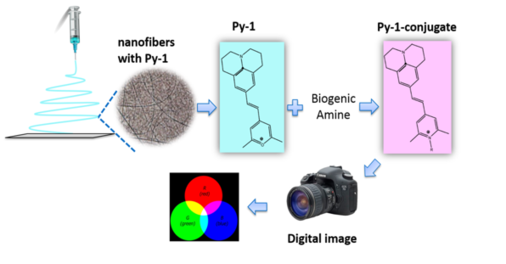
New Materials and Tools for Optical Chemical Sensors
New materials are key tools to promote the properties of optical chemical sensors and test stripes. We recently showed sensor microplates to be useful tools because they save sample and reagent volume, enable rapid readout and can be rendered reusable. Another concept is to use electrospun nanofibers in optical sensing. These fibers are an emerging material in biosensing and chemosensing because they have a high porosity, offer a high surface-to-volume ratio and an immense surface area, can be mass-produced and can be reusable.
Selected publications:
- Yurova NS, Danchuk A, Mobarez SN, Wongkaew N, Rusanova TY, Baeumner AJ, Duerkop A, 2018, Functional Electrospun Nanofibers for Multimodal Sensitive Detection of Biogenic Amines in Food via a Simple Dipstick Assay, Anal Bioanal Chem, 410:1111-1121, DOI: 10.1007/s00216-017-0696-9.
- Azab HA, El-Korashy SA, Anwar ZM, Khairy GM, Steiner M-S, Duerkop A, 2016, Validation of a Sensor Microtiter Plate for Fluorescent Determination of Biogenic Amines in Meat and Cheese, J Fluoresc 26:1905-1916, DOI: 10.1007/s10895-016-1885-1.
- Burmistrova NA, Meier RJ, Schreml S, Duerkop A, 2014, Reusable Optical Sensing Microplate for Hydrogen Peroxide Using a Fluorescent Photoinduced Electron Transfer Probe (HP Green), Sens Actuat B 193:799-805, doi: 10.1016/j.snb.2013.12.025.
Methods and Expertise
We use most techniques of optical molecular spectroscopy and many of them in microplates. We cover reflectometry, photometry, polarimetry, circular dichroism and all parameters of luminescence (intensity, polarization, lifetime, delayed luminescence, electrochemiluminescence and chemiliminescence. Optical sensors are measured continuously in flow-through, via fiber optics or in microplates. Reading of DNA and protein arrays on glass and other substrates with a laserscanner is also offered.
Ion analyses can performed optically and can be are validated by atomic spectroscopy. For this purpose, we are running an inductively-coupled plasma atomic emission spectroscopy instrument (Spectroflame) and an inductively-coupled plasma mass spectrometer (PE Elan 9000). The atomic spectroscopy facilities are also available for all groups of the faculty and of the university that require trace element quantitation.
Selected publication:
- Assmann N, Dettmer K, Simbuerger JMB, Broeker C, Nuernberger N, Renner-Sattler K, Courtneidge H, Klootwijk ED, Duerkop A, Hall A, Kleta R, Oefner PJ, Reichold M, Reinders J*, 2016, Renal Fanconi´s Syndrome Is Caused by a Mistargeting-Based Mitochondriopathy, Cell Rep 15:1423-1429, DOI: 10.1016/j.celrep.2016.04.037.
Prof. Dr. Axel Dürkop
Institute of Analytical Chemistry,
Chemo- and Biosensors
Phone 0941/943-4063
Telefax 0941/943-4064
E-Mail: axel.duerkop@ur.de



