CV
Scientific Career
- July 2024: Galenus Foundation Guest Professor at the Department of Chemical Engineering, Massachusettes Institute of Technology (Prof. Dr. Robert Langer)
- Since 2023: Co-Editor in Chief of the European Journal of Pharmaceutics and Biopharmaceutics
- Since 2013: Co-Editor of the European Journal of Pharmaceutics and Biopharmaceutics
- 2013: Habilitation in Pharmaceutical Technology, University Regensburg
- since 2012: Akademische Rätin at the Department of Pharmaceutical Technology, University Regensburg
- 2008 and 2009: Postdoctoral fellow at the Department of Chemical Engineering, Massachusettes Institute of Technology (Prof. Dr. Robert Langer)
- 2005 - 2012: Research associate at the Department of Pharmaceutical Technology, University Regensburg
- 2002 - 2005: Graduate student at the Department of Pharmaceutical Technology, University Regensburg (Prof. Dr. Achim Göpferich)
- 1996 - 2001: Undergraduate student at the Institute of Pharmacy, Ruprecht-Karls-University Heidelberg
Publications
Publication List
The following list is a selection of all publications of apl. Prof. Dr. Miriam Breunig (articles, book chapters, theses and patents published 2008 and later). You can find the full publication list on the publication server of the University.
Publication list of apl. Prof. Dr. Miriam Breunig
Research
The Breunig group develops innovative drug delivery systems for small drug molecules and biologics such as proteins and nucleic acids. The team works on the following topics:
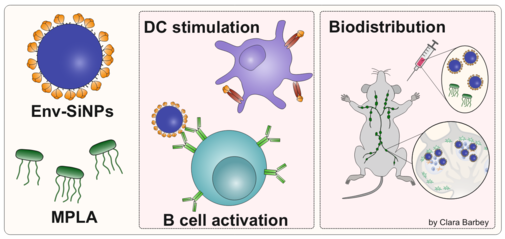
Development of a nanoparticulate HIV vaccine
Delivery of antigens to draining lymph nodes is essential for a potent HIV vaccine. Therefore, we use silica nanoparticles (SiNPs) as delivery platform for the multimeric presentation of HIV´s Envelope (Env) immunogen. We optimize the site-specific coupling reaction to achieve virus-mimetic nanoparticles that are physico-chemically characterized regarding size, zeta potential and functionality of the antigen. The multivalent antigen presentation leads to an increased avidity to B cell receptors followed by a higher B cell activation. Together with our partners from the HIVacToGC consortium we also investigate the kinetics of Env delivery to the lymph nodes in vivo.
The SiNPs are a platform technology which is also transferred to other protein-based antigens like e.g. the receptor binding domain (RBD) of SARS-CoV-2.
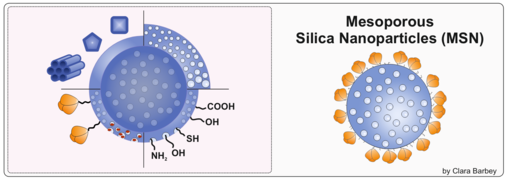
Mesoporous silica nanoparticles for the delivery of antigens and adjuvants
Mesoporous silica nanoparticles have (MSN) an extremely high specific surface area and thus the advantage over SiNPs that their degradation time can be tuned by the pore size. Together with the group of Prof. Mika Lindén were explore MSNs as delivery systems for antigens and adjuvants.
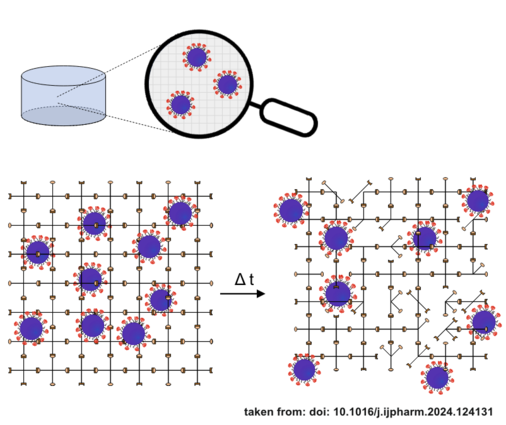
Hydrogels for sustained release of nanoparticulate antigens
Immunization usually requires several visits to a doctor to receive full immune protection. However, this conventional prime boost vaccination regimen is not as efficient regarding eliciting an immune response compared to other kinetics of vaccine application. Therefore, we create hydrogels that release nanoparticulate vaccines with various kinetics over a prolonged time.
Nanoparticulate drug delivery systems for the treatment of primary open-angle glaucoma
Primary open-angle glaucoma is a neurodegenerative disease of the optic nerve and one of the leading causes of blindness worldwide. Pathologic changes in the anterior chamber of the eye are critical in the development of primary open-angle glaucoma because aqueous humor outflow is obstructed by the trabecular meshwork and Schlemm's canal. Conventional treatment with eye drops does not bring the sweeping success due to an extremely low bioavailability after topical application. In addition, the applied active substances do not intervene at the root cause of the disease. We are developing nanoparticulate drug delivery systems that transport active substances to the site of action in the trabecular meshwork and Schlemm's canal after intracameral application. Furthermore, the applied drugs like small interfering RNA directly interfere with the pathomechanism of primary open angle glaucoma and do not only act on symptoms of the disease. This project is a cooperation with Prof. Rudolf Fuchshofer .

Figure taken from doi: 10.1002/smll.201803239
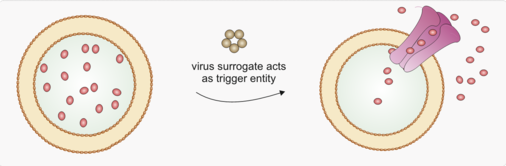
Tools to gain spatial and temporal control of drug release
Together with our collaboration partners (Prof. Dr. Antje Bäumner, Prof. Dr. Diana Pauly, Prof. Dr. Ralf Wagner, Microcoat Biotechnology GmbH) we are developing a liposome-based assay for the quantification of SARS-CoV-2 neutralizing antibodies in patient sera. Our task in the consortium is to develop a virus surrogate which acts as trigger entity to release indicator molecules from liposomes.