
liposomes
Liposomes ha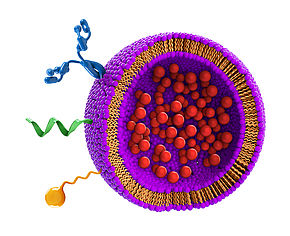 ve been developed in our research group as bioanalytical tools for signal generation and instantaneous signal amplification based on the early work in Dr. Richard Durst’s group. We have established protocols that allow for reliable liposome synthesis and for physical, chemical and biological characterization. Comparing lipos
ve been developed in our research group as bioanalytical tools for signal generation and instantaneous signal amplification based on the early work in Dr. Richard Durst’s group. We have established protocols that allow for reliable liposome synthesis and for physical, chemical and biological characterization. Comparing lipos
omes to other signal generation and amplification means such as enzymes, quantum dots, gold nanoparticles, and fluorescence molecules had demonstrated their superior performance for bioanalytical sensors. We also study liposome stability under extreme conditions including dehydration, lyophilization, and storage at temperatures above 40 °C, in blood, urine and other matrices. We have investigated liposomes for nucleic acid, protein, and whole cell detection, and their use for bioimaging and for multiplexing. New projects investigate the development of multimodal liposomes to further enhance their bioanalytical capabilities.
For example:
- Rink, S., Kaiser, B., Steiner, M.-S., Duerkop, A., Baeumner, A.J. “Highly sensitive interleukin 6 detection by employing commercially ready liposomes in an LFA” Analytical and Bioanalytical Chemistry, (2022) 414: 3231–3241, https://doi.org/10.1007/s00216-021-03750-5
- Yagati, A.K., Behrent, A., Tomanek, V., Chavan, S.G., Go, A., Park, S.R., Jin, Z., Baeumner, A.J., Lee, M. “Polypyrrole-palladium nanocomposite as a high-efficiency transducer for thrombin detection with liposomes as a label” Analytical Bioanalytical Chemistry (2021) 3205 - 3217, https://doi.org/10.1007/s00216-021-03673-1
- Hofmann, C., Kaiser, B., Märkl, S., Duerkop, A., Baeumner, A.J. ‚ Cationic liposomes for generic signal amplification strategies in bioassays, Analytical and Bioanalytical Chemistry, 412(14), 3383-3393. Front cover, https://doi.org/10.1007/s00216-020-02612-w
- Hermann, C., Hofmann, C, Duerkop, A., Baeumner, A.J. “Magnetosomes for bioassays by merging fluorescent liposomes and magnetic nanoparticles: encapsulation and bilayer insertion strategies“ Analytical and Bioanalytical Chemistry (2020), 412, 6295–6305, https://doi.org/10.1007/s00216-020-02503-0
- Mayer, M., Takegami, S., Neumeier, M., Rink, S., Jacobi von Wangelin, A., Schulte, S., Vollmer, M., Griesbeck, A.G., Duerkop, A., Baeumner, A.J., “Electrochemiluminescence Bioassays with a Water-Soluble Luminol Derivative Can Outperform Fluorescence Assays” Angew. Chem. Int. Ed. (2018), 57, 408-411, DOI: 10.1002/anie.201708630
- Fenzl, C., Hirsch, T., Baeumner, A.J. “Liposomes with High Refractive Index Encapsulants as Tunable Signal Amplification Tools in Surface Plasmon Resonance Spectroscopy” Anal. Chem., 2015, 87 (21), pp 11157–11163, DOI: 10.1021/acs.analchem.5b03405
- Edwards, K.A., Baeumner, A.J. “Enhancement of Heterogeneous Assays using Fluorescent Magnetic Liposomes” Anal. Chem. vol 86 (13), pp. 6610 – 6616 (2014), DOI: 10.1021/ac501219u
- Edwards, K.A., Meyers, K. J., Leonard, B., Baeumner, A.J. “Superior performance of liposomes over enzymatic amplification in a high-throughput assay for myoglobin in human serum” Anal. Bioanal. Chem. vol. 405(12), pp. 4017-4026 (2013), DOI: 10.1007/s00216-013-6807-3
- Edwards, K.A. and Baeumner, A.J. “DNA-Oligonucleotide Encapsulating Liposomes as a Secondary Signal Amplification Means” Analytical Chemistry, vol. 79(5), pp. 1806 – 1815 (2007), DOI: 10.1021/ac061471s



