A1: Temporospatial changes of TSPO expression in glioblastoma in response to therapy and during disease progression – a multimodal TSPO PET, amino acid PET and MRI study
Nathalie Albert, Louisa von Baumgarten
Temporospatial changes of TSPO expression in glioblastoma in response to therapy and during disease progression – a multimodal TSPO PET, amino acid PET and MRI study
TSPO is overexpressed in primary brain tumors and has been shown to correlate with tumor malignancy and a poor prognosis. Our previous work performed in the first funding period has shown that the PET signal of gliomas in TSPO PET and amino acid PET ([18F]FET) are different and also differ from contrast enhancement in MRI. Furthermore, PET signals may change in response to therapy and during disease progression and its impact on the clinical outcome and individual prognosis of glioblastoma patients has not been assessed so far.
Therefore, this project aims to visualize temporal and spatial changes of TSPO expression by means of a single arm observational study using longitudinal multimodal TSPO / [18F]FET PET and ceMRI for patients with glioblastoma, and to correlate imaging findings with changes on the cellular and molecular level as well with the clinical outcome.
In work package 1 patients with newly diagnosed brain tumors suspicious for glioblastoma are prospectively enrolled in an observational study. All data retrieved from clinical workup, clinical-follow-up, imaging and neuropathological analysis are collected. Tumor specimen retrieved from tumor resection or stereotactic biopsies are spatially correlated with PET and MRI data, which enables a precise comparison of imaging information with histopathological findings and pathway analyses for project A2/3. This collaboration with project A2/A3 allows in-depth analyses of changes of the tumor microenvironment, cellular contributions to the TSPO signal (myeloid cells vs. tumor cells) and molecular alterations in a unique longitudinal study cohort with tissue samples obtained with TSPO PET image guidance at primary diagnosis and at relapse.
Work package 2 focusses on advanced image analysis of the multimodal imaging data and its correlation with clinical and neuropathological data obtained in work package 1. The analyses include pharmacokinetic modelling, machine learning and deep learning approaches to perform non-invasive tissue classification, distinguish therapy responders from non-responders at an early time point and to predict the time and localization of tumor recurrence and ultimately, to link imaging features with overall survival.
A2
A3
B1: Nuclear magnetic resonance (NMR) spectroscopy of human TSPO
Markus Zweckstetter
Nuclear magnetic resonance (NMR) spectroscopy of human TSPO
The three-dimensional structure of mouse TSPO (mTSPO) was previously determined by my lab using solution-state NMR spectroscopy. In addition, we studied the influence of a clinically relevant polymorphism in mTSPO, which results in the substitution of alanine by threonine at position 147 and decreases the affinities of many radioligands and therapeutics, on the mTSPO structure. We also found out that mTSPO is more flexible when no high-affinity radioligand is bound to the protein. A further focus of our studies was the analysis of the interaction of mTSPO with cholesterol using solid-state NMR spectroscopy. The three-dimensional structure of human TSPO is, however, unknown. In this project, we thus propose to study the structure of human TSPO (hTSPO) using solution- and solid-state NMR spectroscopy. To this end, we will first – using a model protein – perform tests on how to optimize the NMR process of resonance assignment and structure determination of alpha-helical membrane proteins. Subsequently, we will apply this methodology to hTSPO in the absence and presence of high-affinity ligands that are studied within the research unit (such as XBD173). This work will address the question if hTSPO folds – in the absence of hTSPO-specific ligands – into a stable conformation when it is solubilized in either detergents or lipids with distinct properties. Subsequent steps will be the determination of the structure of hTSPO, analysis of the molecular details of its interaction with selected high-affinity ligands and the influence of the A147T polymorphism on these interactions.
C2: Biological functions of glial TSPO in retina and brain models of the cholesterol storage disease Niemann-Pick type C (NPC)
Antje Grosche, Sabina Tahirovic
Biological functions of glial TSPO in retina and brain models of the cholesterol storage disease Niemann-Pick type C (NPC)
Translocator protein 18 kDa (TSPO) is an integral protein of the outer mitochondrial membrane. Although its cellular functions are still debated, it is thought to play a role in neurosteroidogenesis, mitochondrial function, cell metabolism and neuroinflammation. In the central nervous system (CNS), TSPO expression is predominantly found in ependymal cells, microglia, and astrocytes, but its cellular expression pattern largely changes under neuroinflammatory and neurodegenerative conditions. For the retina, TSPO was reported to be a regulator of cholesterol and neurosteroid metabolism, so this part of the CNS can serve as an ideal and, most importantly, easy-to-study model to analyze cell-type-specific beneficial or detrimental effects of TSPO. In the healthy tissue, we detected the highest TSPO expression in Müller cells, the major macroglia in the retina. In contrast, in retinal pathologies, TSPO is strongly upregulated by reactive microglia. Our research project specifically aims to compare the function of glial TSPO in the retina with results from corresponding cell types in the brain. To determine the function of TSPO under physiological and neurodegenerative conditions, we selected the Niemann Pick type C (NPC) metabolic disease model with distinct retinal and brain pathology. This monogenetic disease – predominantly caused by the loss of NPC1 function – is characterized by cholesterol storage and metabolic defects. In our new microglia-specific NPC1 knockout model, we found early cell autonomous defects of microglia and massive upregulation of microglial TSPO. These microglial deficits resulted in pathological changes in other cell types such as astrocytes and neurons in various CNS regions, including the retina. Therefore, this model provides a unique opportunity to study the functions of TSPO in glial cholesterol transport, neurosteroidogenesis, neuroinflammation, and cellular metabolism in the retina and brain. Thus, we aim to clarify whether an increase in glial TSPO triggers protective or detrimental responses in the CNS and how this may contribute to neuropathology. In a translational approach, we will analyze the effect of TSPO loss on glial functions including cholesterol metabolism and neurosteroid biosynthesis in human retinal organoids with and without microglia under homeostatic and NPC conditions as done in our mouse model. These comparative studies of TSPO signaling cascades in murine and human glia will help to significantly expand our understanding of TSPO biology and its relevance to human disease.
C3
C4: Interactions of brain TSPO and stress-related systems under acute and chronic stress conditions
Inga Neumann, Oliver Bosch
Interactions of brain TSPO and stress-related systems under acute and chronic stress conditions
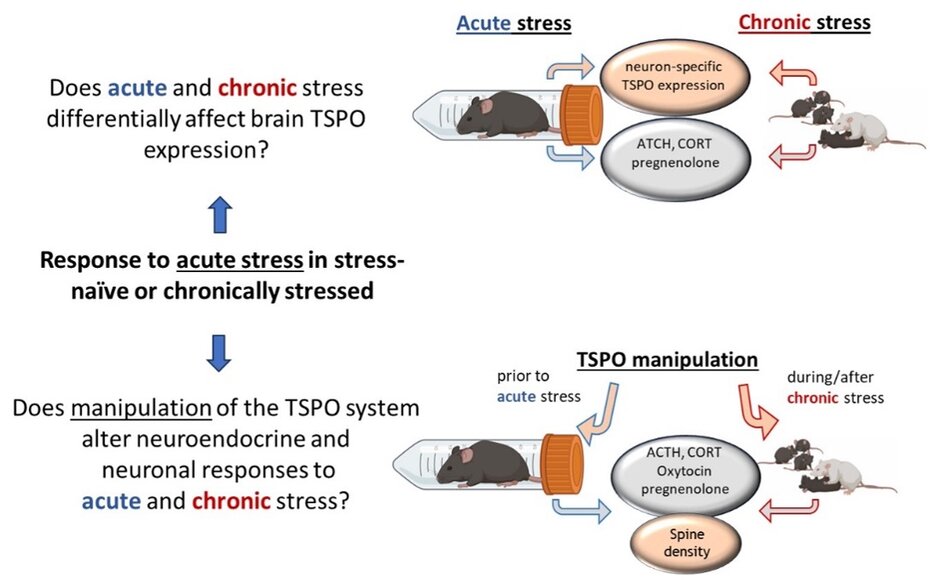
Stress-related psychiatric disorders such as anxiety, depression, and post-traumatic stress disorder (PTSD) remain major unmet clinical challenges. Increasing evidence identifies the translocator protein 18 kDa (TSPO) as a promising neurobiological target for novel treatment strategies in neuropsychopharmacology. TSPO facilitates cholesterol transport into mitochondria and thereby regulates neurosteroidogenesis. Neurosteroids act as potent allosteric modulators of GABAa receptors, which are critically involved in anxiety regulation and hypothalamo–pituitary–adrenal (HPA) axis function.
Clinically used TSPO ligands such as etifoxine exert robust anxiolytic effects comparable to benzodiazepines, yet with a distinct pharmacological profile. Importantly, TSPO expression is altered in patients with stress-related disorders, as shown by PET imaging studies. However, TSPO levels may also change dynamically in response to neuronal activation, underlining the need for mechanistic studies that integrate stress exposure, TSPO regulation, and behavioral outcomes.
In the first funding period we studied TSPO in our well-established animal models of emotionality and stress. Wistar rats selectively bred for high (HAB) or low (LAB) anxiety-related behavior represent a validated genetic model of pathological anxiety, depression-like behavior, altered stress coping, and dysregulated HPA axis activity. The clinically available TSPO ligand etifoxine was systematically investigated across sexes and models. Etifoxine reduced anxiety-related behavior specifically in female HAB rats, indicating sex- and trait-dependent efficacy. While acute antidepressant-like effects were not observed, etifoxine normalized reduced hippocampal spine density in female HAB rats, suggesting beneficial effects on neuronal plasticity. Moreover, etifoxine induced region- and line-specific changes in oxytocin receptor binding, particularly within the lateral septum and central amygdala, pointing to interactions between TSPO, neurosteroids, and social–emotional circuits. Importantly, effects on HPA axis reactivity were highly context-dependent: prolonged treatment enhanced stress-induced corticosterone release in non-anxious rats, whereas anxiety-prone animals remained unaffected (Fischer et al., 2025).
In the second funding period we aim to advance these findings through a systematic, back-translational approach. The project focuses on bidirectional interactions between stress exposure and TSPO function, examining how acute versus chronic stress alters TSPO expression in discrete brain regions and cell populations, from glial cells to neurons, and how pharmacological TSPO manipulation affects behavioral, neuroendocrine, and cellular stress markers. Using acute restraint stress and the chronic subordinate colony housing (CSC) model, a chronic psycho-social stressor, combined with behavioral testing, hormone measurements, neurosteroid analysis, and receptor mapping, the project seeks to identify stress- and sex-specific TSPO-dependent mechanisms.
By elucidating how TSPO coordinates neurosteroid signaling, HPA axis activity, and emotional behavior under different stress conditions, this work aims to define biologically grounded targets for translational research. Ultimately, the findings may inform the rational clinical use of TSPO ligands such as etifoxine and help optimize treatment strategies for stress-related mental disorders.
C5: Differential effects of TSPO ligands on functional connectivity and metabolism related to fear and anxiety in the human brain
Caroline Nothdurfter, Jens Schwarzbach, Rainer Rupprecht
Differential effects of TSPO ligands on functional connectivity and metabolism related to fear and anxiety in the human brain.
TSPO ligands in the treatment of depression: proof-of-concept and underlying mechanisms of action.
TSPO ligands in the treatment of depression: proof-of-concept and underlying mechanisms of action.
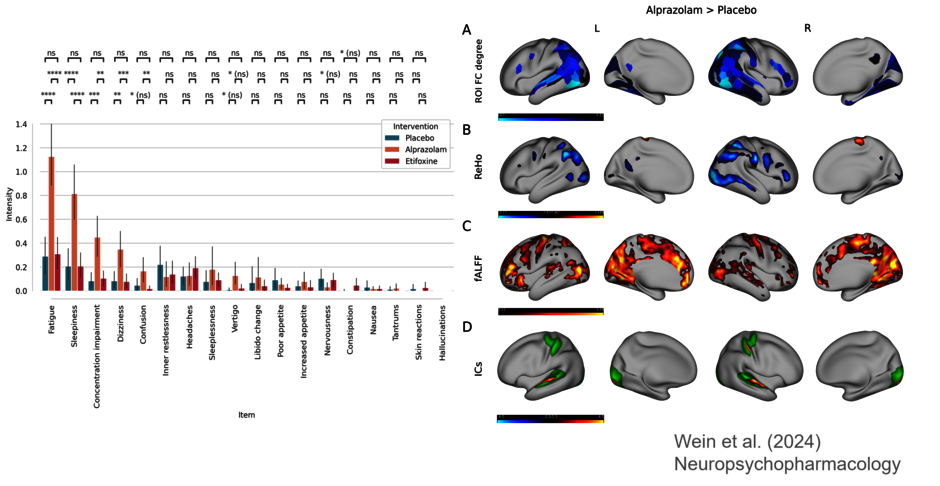
Benzodiazepines are fast acting anxiolytics which primarily target synaptic GABAA receptors. However, common side effects are sedation, tolerance development, and abuse liability. TSPO ligands promote endogenous neurosteroidogenesis which modulate extrasynaptic GABAA receptors. Meanwhile, exogenous neurosteroids have been shown to exert rapid antidepressant effects in postpartum depression and major depressive disorder (Rupprecht et al. 2022, 2026; Riebel et al. 2025). To delineate putative differences between bezodiazepines and TSPO ligands, in the first funding period we perfomed a clinical study in 36 healthy male volunteers to compare the effects of the TSPO ligand etifoxine with the benzodiazepine alprazolam regarding anxiolytic efficacy, modulation of GABergic excitability, modulation of neuronal networks, and microbiome composition. In the second funding period we conducted a placebo controlled clinical proof of concept study in patients with major depression to evaluate whether the TSPO ligand etifoxine exerts rapid antidepressant effects similar to exogenous neurosteroids such as zuranolone (Rupprecht et al. 2022, 2026). Moreover, we investigated the impact of etifoxine on cognition and depression related neuronal networks (Brunner et al. 2024).
In the first funding period we showed that alprazolam had more pronounced GABAergic effects than etifoxine in a double pulse TMS paradigm and produced more sedation (Riebel et al. 2023). Moreover, sedation induced by alprazolam disconnects low-level resting state functional networks but simultaneously increases their within-network connectivity (Wein et al. 2024). Moreover, we showed additional effects of alprazolam-associated sedation using our inhouse developed fusion searchlight (FuSL) framework, which integrates complementary information from multiple resting-state fMRI metrics, thereby enhancing the accuracy of pharmacological treatment prediction from rs-fMRI data (Wein et al. 2025). Treatment with etifoxine reduced the abundance of a few bacterial species, which are currently seen as beneficial components of a healthy intestinal microbiome (Manook et al. 2023), whereas absolute plasma neurosteroid levels were not affected (Riebel et al. 2025). In a task-based paradigm, alprazolam reduced subjective nervousness more prominently than etifoxine, while no deprsignificant effect of either medication could be detected by means of fMRI (Riebel et al. in preparation).
In the second funding period we conducted a clinical proof of concept study in 50 patients with major depressive disorder to assess whether adjunct treatment with the TSPO ligand etifoxine may fasten the clinical response to antidepressant treatment via enhancement of endogenous neurosteroidogenesis (Brunner et al. 2024). Moreover, a putative impact on cognition is inverstigated by a cognitive test battery. Finally, the investigation of depression related neuronal networks will provide insight into the mechanisms of action of putative antidepressant effects of TSPO ligands. The recruitment for the study is completed now. The respective data analyses are under way.
